Measuring the Threat of Nipah Disease
The Nipah virus originates from large fruit-eating bats. Even though the threat level of Nipah disease is moderate and the spread of the disease is limited, we still have to anticipate it.
This article has been translated using AI. See Original .
About AI Translated Article
Please note that this article was automatically translated using Microsoft Azure AI, Open AI, and Google Translation AI. We cannot ensure that the entire content is translated accurately. If you spot any errors or inconsistencies, contact us at hotline@kompas.id, and we'll make every effort to address them. Thank you for your understanding.
The following article was translated using both Microsoft Azure Open AI and Google Translation AI. The original article can be found in Menakar Ancaman Penyakit Nipah

Illustration
Kompas on September 23 2023 reported the fourth nipah disease outbreak in Kerala, India. At the end of August 2023, six people were infected, two of whom died. In the first case (2018), nipah disease killed 10 people. In 2001-2019, Hauser et al (2021) recorded four cases of nipah disease in India with 92 cases, 68 people died (case fatality rate 73.9 percent).
Before it was discovered in India, nipah disease was discovered in Bangladesh (1998) in people who drank sap from palm trees. It is suspected that sap is contaminated with the Nipah virus through saliva, urine or feces from fruit-eating bats. Luby et al (2006) reported that in 2004-2005, 12 cases of nipah disease were found, 11 of whom died (CFR 92 percent). Lubby et al included Bangladeshi nipah disease as food-borne transmission, different from Malaysian nipah.
Antibodies and the Nipah virus have been found in bats in Thailand (2002-2005), but there have been no reported cases in humans.
Also read: Recurring Nipah Outbreaks in India and the Risks in Indonesia
The nipah virus first broke out in Nipah, Perak, Malaysia in September 1998. Two months later, it spread to Negeri Sembilan. A total of 265 people were affected, with 105 fatalities (CFR 38.5 percent). In Singapore, 11 cases of nipah virus were found during pig slaughtering in 1999, with one death (Patton et al., 1999). The pigs had been imported from Malaysia.
Considering that the nipah disease is present around our country, how big of a threat is this disease?
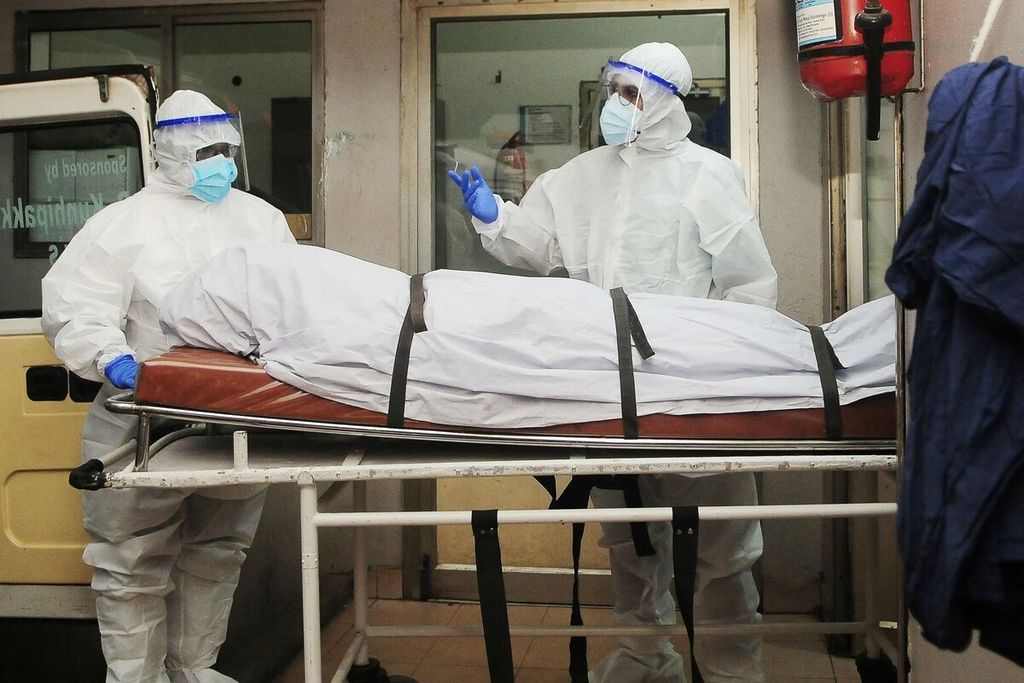
Health workers, Tuesday (12/9/2023, used personal protective equipment (hazmat) when carrying the body of a suspected Nipah virus infection at a hospital in Kozikode, Kerala, India.
History
Initially, the Nipah disease affected pigs in Nipah, Malaysia. The clinical symptoms were pronounced in young pigs, such as respiratory problems, while adult pigs experienced neurological disorders. The morbidity rate among pigs was 100 percent, but the mortality rate was relatively low (1-5 percent).
Not long afterward, workers at the pig slaughterhouse became ill. They are infected from the blood or organs of pigs when they are slaughtered. It's easy to guess, this disease is zoonotic. Because the disease was considered malignant in humans, specimens in the form of pig tissue and recovered pig serum were sent to the AAHL (Australian Animal Health Laboratory) laboratory in Geelong, Australia, (now called the Australian Center for Disease Preparedness/ACDP) which has a high security level (BSL4).
Pig serum that has been cured has a cross-reaction with the Hendra virus that causes the death of horses and several people in Australia. For the time being, the virus is called Hendra-Like Virus. The Hendra virus belongs to the family Paramyxoviridae, originating from fruit-eating bats.
The Nipah virus, like the Hendra virus, originates from large fruit-eating bats of the Pteropodidae family.
Malaysia succeeded in isolating the virus that causes the disease, but for identification the virus isolate was sent to the American CDC. The virus was successfully identified as the Paramyxovirus group. A joint investigation involving AAHL Australia, the Collaborating Center for Tropical Diseases Japan, and the American CDC stated that the virus that causes the disease is a new virus so it was named after the place where the disease was first discovered, namely Nipah.
To free themselves from the nipah disease, more than 1 million pigs were destroyed (1999). Until now, there have been no reports of nipah disease in Malaysia.
The Nipah virus, like the Hendra virus, originates from large fruit-eating bats of the Pteropodidae family. This bat is widespread from Australia, Southeast Asia, South China, India and Madagascar.
McLean et al (2022) said that pigs are amplifier hosts for the Nipah virus. This means that after entering the pig's body, the virulence of the Nipah virus increases, causing quite a number of severe cases in people with a fairly high mortality rate.
The vaccine for Nipah disease is still under research and is not yet available in the market.
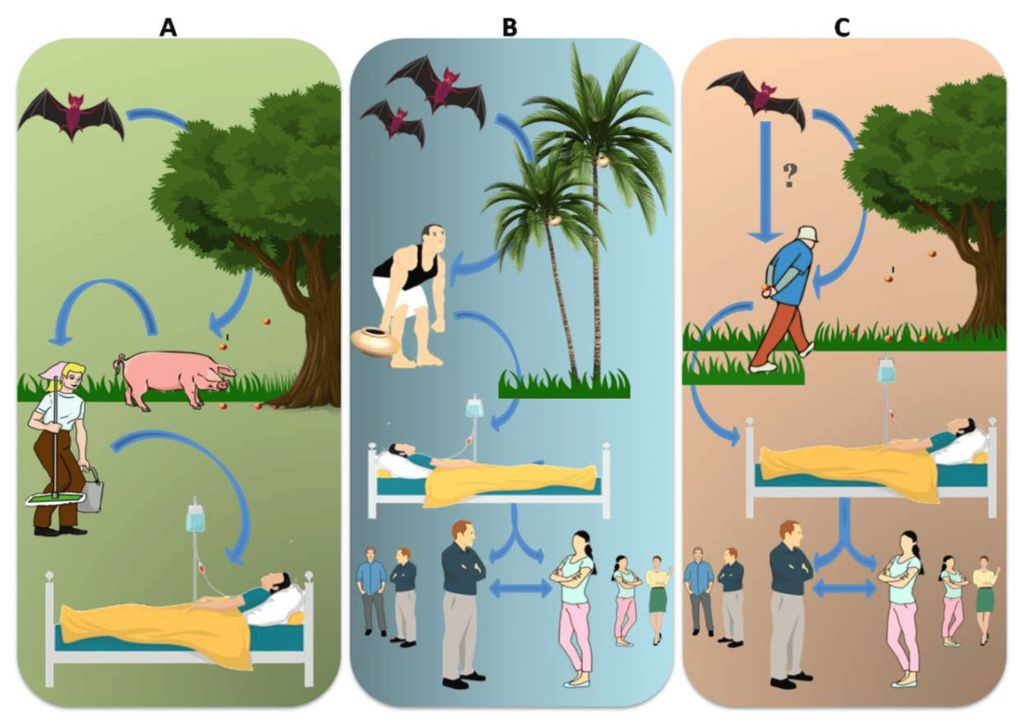
The transmission route of the Nipah virus (NiV) varies by location. (A) In Malaysia, fruit bitten by NiV-M contaminated bats is consumed by pigs, and workers handling these pigs are infected with NiV-M. (B) In Bangladesh, consumption of palm sap contaminated with bat saliva and excrement causes NiV-B infection in humans and spreads further through nosocomial means. Infected bats excrete the virus through their urine, droppings, and saliva. (C) In India, direct transmission from bats to humans has been reported in the state of Kerala, but this is not supported by adequate evidence. Nosocomial spread of NiV-B has been reported in two different states - Kerala and West Bengal. Source: Vinod Soman Pillai et al. (Journal MDPI, 2020)
Medium threat level
Nipah virus antibodies have been detected in bats in North Sumatra, West Kalimantan, North Sulawesi, West Java, and East Java, but not in pigs (Sendow et al., 2006). This indicates that the Nipah virus is circulating in bats but has not yet spread to pigs. It is predicted that the entry of the Nipah disease into Indonesia is only a matter of time.
Regions with high pig populations have the highest risk of transmission. Pigs can contract the Nipah virus through consuming leftover fruit that has been bitten by bats. The fruit is contaminated with the Nipah virus through bat saliva. Similar occurrences happen with horses that contract the Hendra virus in Australia.
Also read: Vigilance and Surveillance Strengthened to Anticipate Transmission of the Nipah Virus
Learning from the nipah case in Bangladesh, tappers need to be careful as the sap they tap from trees can be contaminated with the Nipah virus. The likelihood of transmission among tappers in Indonesia is lower than among pig cutters.
Early detection of nipah disease in pigs can reduce the potential for transmission to humans. By strictly isolating patients, transmission can be avoided.
From the above observation, the threat level of Nipah disease is moderate. The spread of the disease is limited as transmission between humans is relatively difficult. The challenge for Indonesia would be the elimination of pigs in the affected areas, which requires a large amount of compensation funds.
From the description above, we need to be cautious about the nipah disease, but there is no need to panic as laboratories are readily equipped for early detection in animals and patient isolation facilities are available.
Soeharsono, Former Investigator of Animal Diseases

Soeharsono